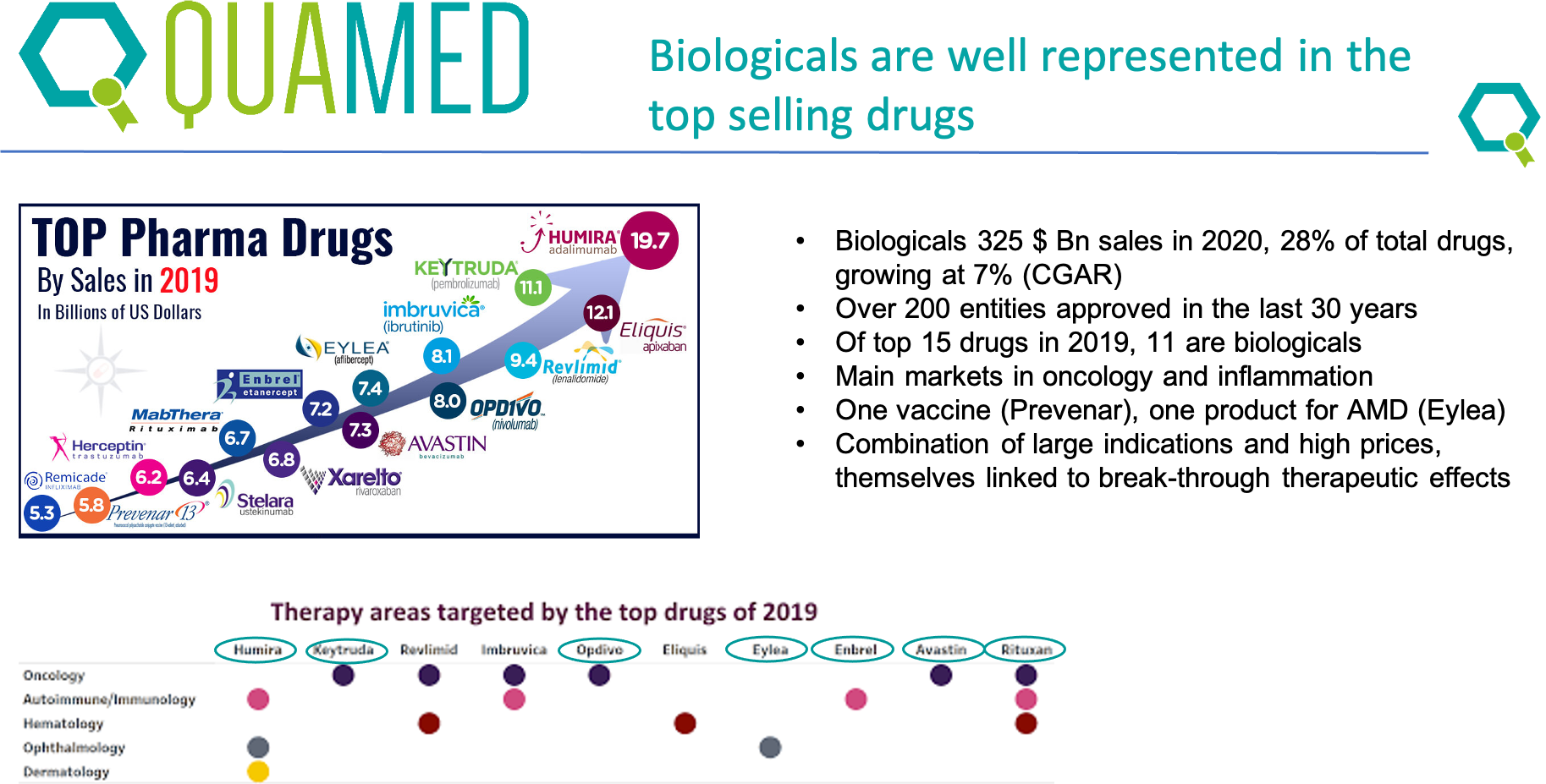
This week QUAMED is organising a webinar on biological medicines. What are they, how are they made, how are they regulated and what about access to biological medicines in LMIC settings. The webinar is presented by Thomas Chattaway and it is given in English and in French. Accessible for all our members.
Search this website
All topics
- Access to medicines (43)
- Alerts and recalls (7)
- Audits & Inspections (2)
- BPF (1)
- BPSD (2)
- ENABEL (6)
- Falsified and sub-standard medicines (10)
- GSDP (2)
- International (10)
- ITM (1)
- Latin America (1)
- Literature updates (32)
- MQAS (11)
- National regulatory authority (3)
- News (33)
- PAHO (2)
- Podcast (2)
- QA standards (13)
- QCP (11)
- Quality assurance (33)
- Quamed News (253)
- Recruitment (9)
- Research (7)
- Technical visits (2)
- Training (8)
- USAID (5)
- Webinaire (1)
- Webinar (23)
- WHO (28)